NNB Nutrition announces self-affirmed GRAS status of its dihydroberberine
NNB Nutrition’s GlucoVantage®, which is called Dihydroberberine (DHB), has achieved self-affirmed GRAS status, NNB has demonstrated that DHB is safe and useful in added doses in foods and beverages. This means that DHB can be considered safe for intended uses in food and beverages, including nutrition bars and nutrition drinks such as energy drinks or milk-based drinks. DHB has also been carefully characterized and is free of allergens, pesticides and other toxic contaminants.
In the independent GRAS status conclusion for DHB, the GRAS Panel considered the preclinical toxicity studies and clinical trial in humans to be the key information supporting the conclusion that DHB is not expected to produce adverse effects. adverse effects on human health under the intended conditions of use. . Evidence supporting the safety of DHB was obtained from the safety data on berberine. Based on the data above and the information presented here, NNB has concluded that the intended uses of DHB when used as an ingredient at a maximum use level of 55g per bar per serving (300mg/day) in nutrition/cereal bars, and up to 325 ml per drink per serving (300 mg/day) in nutrition drinks is GRAS based on scientific procedures. The general recognition of the NNB GRAS determination is supported by the unanimous consensus rendered by an independent GRAS panel, qualified by experience and scientific training to evaluate proposed uses of DHB. This statement is made in accordance with the FDA Standard for Food Ingredient Safety.
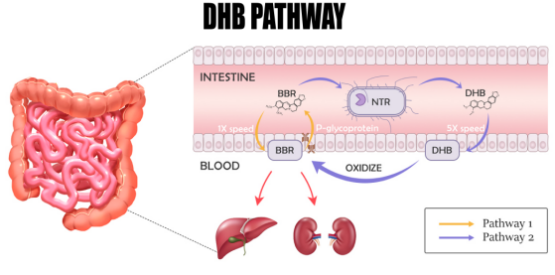
GlucoVantage® is the first commercially available brand of dihydroberberine (DHB), which is a patented active metabolite of berberine. Berberine has been shown to optimize blood sugar, improve insulin sensitivity and body composition. Dihydroberberine solves the limitations of berberine when it comes to low bioavailability and gastrointestinal distress. DHB is one of 17 active metabolites of berberine, which is produced naturally in the body via a reduction process that occurs after ingestion of berberine by gut microbes. Dihydroberberine is then converted back to berberine after absorption through the intestines. Dihydroberberine supplementation may therefore suppress this rate-limiting step in microbial reduction and alleviate the potential gastrointestinal upset that some experience with berberine supplementation. This gastrointestinal distress is likely the result of poor absorption and/or the microbial reduction process.
NNB GlucoVantage® Benefits
1. Less potential gastrointestinal distress than berberine, reducing adiposity and improving insulin sensitivity at lower doses
2. Highly bioavailable form of berberine (up to 5 times more effective)
“With our GRAS certification, we believe this powerful cutting-edge ingredient will be a huge hit in the dietary supplement industry.” said Kylin, the founder of NNB Nutrition.
About NNB
NNB is the leading ingredient-focused biotechnology company employing over 100 research scientists from over 10 countries who research, design and deliver unique ingredients to industry to maximize health, increase performance, increase longevity and improve the appearance. NNB promotes partnerships with companies in the fields of nutraceuticals, pharmaceuticals and medical research.
Media Contact
Company Name: Nanjing Nutrabuilding Biotech Co., LTD. (NNB Nutrition)
Contact person: Amanda
E-mail: Send an email
Call: +86-25 5881 2792
Address:No. 270 Jiqingmen Street, Suning Huigu, Building E6, Room 2105
Town: Nankeen
Country: China
Website: https://www.nnbnutrition.com/